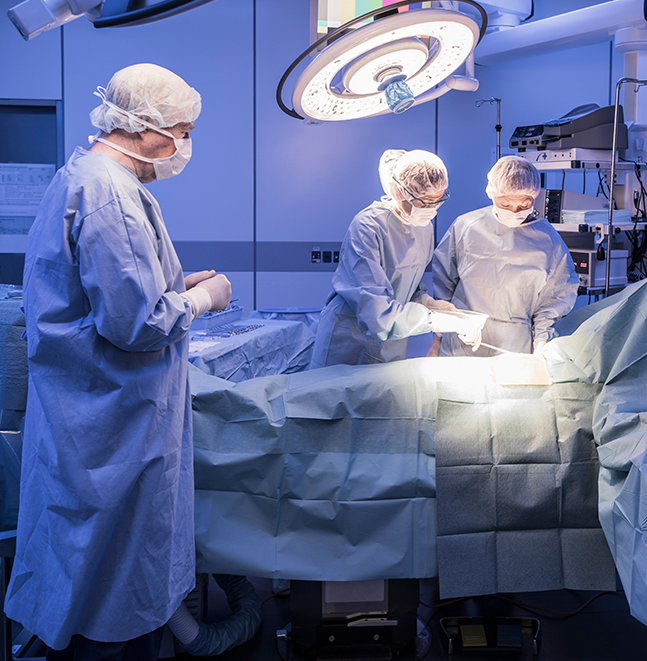
Master & Frank
We'll Ensure You Always get the Best Results
With the beliefs of mastering all business challenges, and being frank with all customers. We own and operate manufacturing plants in mainland China, Cambodia, and Taiwan, products are accredited with the ISO13485,CE (MDR), FDA 510K/NIOSH and GMP/TFDA quality systems.
fun and facts
staff ready to help you
increase the velocity
Accurate and Precisely.
For Services
Medical Products
PRODUCTS
Manufacturing personal protective equipment and medical product.
Surgical Gowns series
EN 13795 – Standard & High Performance
AAMI PB70 – Level 3 & Level 4 Barrier Protection
AAMI PB70 – Level 3 & Level 4 Barrier Protection
Surgical Drapes series
EN 13795 – Standard & High Performance
AAMI PB70 – Level 3 & Level 4 Barrier Protection
AAMI PB70 – Level 3 & Level 4 Barrier Protection
Surgical Packs series
EN 13795 – Standard & High Performance
AAMI PB70 – Level 3 & Level 4 Barrier Protection
AAMI PB70 – Level 3 & Level 4 Barrier Protection
Infection Control series
Isolation gown: meet standard EN13795 (AAMI Level 2 / Level 3/Level 4 barrier provided)
Thump-up PE Gown: Material tested against ASTM F1670 / F1671.
Thump-up PE Gown: Material tested against ASTM F1670 / F1671.
Coverall series
EN 14126:2003/AC:2004 Medical Grade
ASTM PB70 F1670/F1671
EN1149
ASTM PB70 F1670/F1671
EN1149
Face Mask series
ASTM F2100 Level 2/Level 3
EN 14683 Type IIR
FDA 510K/NIOSH 42 CFR Part 84
EN 14683 Type IIR
FDA 510K/NIOSH 42 CFR Part 84
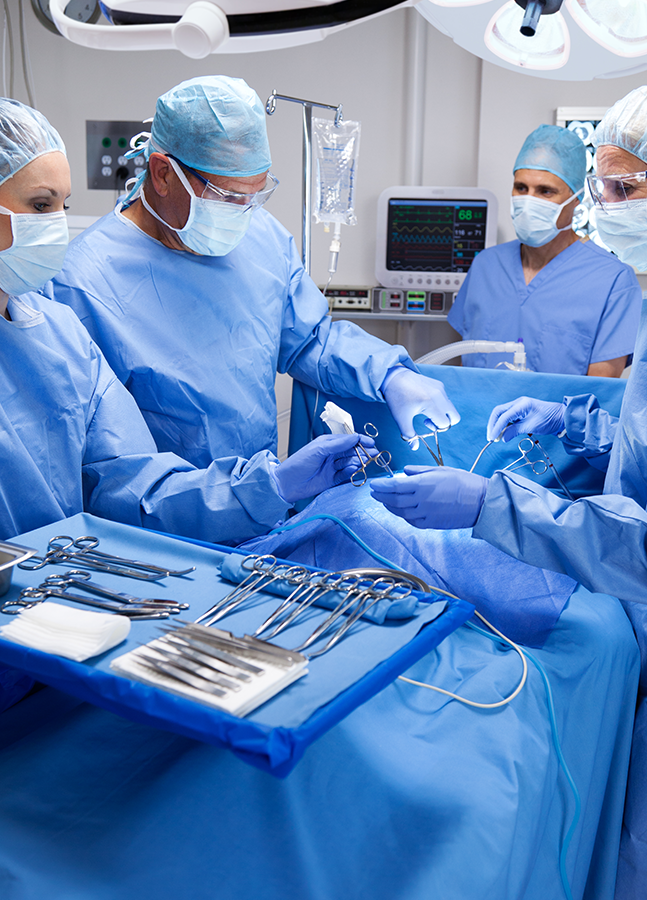
staff ready to help you
OEM/ODM
High Quality Products
Our OEM/ODM Services
Professional Surgical Product Manufacturer
Your Trusted Partner
Surgical Gown Manufacturing
Professional production of high-quality surgical gowns, compliant with ISO13485, CE(MDR), FDA, and 510K international certifications.
Sterilization Services
Professional ethylene oxide sterilization services ensuring the highest safety standards for single-use medical devices.
International Supply
Global distribution network providing reliable and timely supply of surgical gowns to healthcare facilities worldwide.
Product Innovation
Continuous R&D innovation to enhance surgical gown performance and meet evolving medical standards.
Custom Solutions
Tailored surgical gown designs to meet specific requirements of healthcare facilities and medical professionals.
Technical Assistance
Comprehensive technical support and training for healthcare professionals on proper usage and maintenance.
Quality Management
Rigorous quality control processes and comprehensive quality management system implementation.
Quality Certification
Complies with medical device manufacturing standards to meet your market needs.
High Quality Supplier
OUR GOAL
With your supports, we will keep growing.
Our goal is simple:
Do business honestly and meet customers' satisfaction.
Our goal is simple:
Do business honestly and meet customers' satisfaction.
85
%
Technologies
Developed
85
%
High end
Technology

Our Manufacturing Process
Professional Surgical Production
in Four Simple Steps
Requirements Analysis
We begin by understanding your specific needs, including surgical gown specifications, quantity requirements, and regulatory compliance needs for your target market.
Design & Development
Our expert team develops customized surgical gown designs, ensuring optimal protection, comfort, and compliance with international medical standards.
Quality Manufacturing
Production takes place in our state-of-the-art facilities, with strict quality control measures and adherence to ISO13485 and other international standards.
Delivery & Support
Timely delivery of your surgical gowns, complete with necessary documentation and ongoing technical support to ensure your satisfaction.
General FAQ’s
Frequently Asked Questions.
Isolation gowns are essential Personal Protective Equipment (PPE) designed to protect the wearer from exposure to infectious agents, chemicals, and bodily fluids. The choice of material depends primarily on whether the gown is disposable or reusable, and the required level of protection (often classified by AAMI standards, from Level 1 for minimal risk to Level 4 for high risk).
Highly effective bacteria barrier:
Effectively prevents microorganisms from penetrating the non-sterile area into the sterile surgical field.
Waterproof and blood-resistant:
Resistant to the penetration of blood, body fluids, and moisture, which is key to preventing cross-transmission of infection.
Low lint generation:
Low generation of lint or particles to prevent contamination of wounds or surgical instruments.
Antistatic properties: In operating room environments, they must prevent static electricity accumulation and reduce the risk of fire.
Strong and flexible:
Strong and tear-resistant, yet flexible and lightweight for easy laying and storage.
Sterilization:
The material itself must be able to withstand sterilization using gases (such as ethylene oxide) or pressurized steam.
Effectively prevents microorganisms from penetrating the non-sterile area into the sterile surgical field.
Waterproof and blood-resistant:
Resistant to the penetration of blood, body fluids, and moisture, which is key to preventing cross-transmission of infection.
Low lint generation:
Low generation of lint or particles to prevent contamination of wounds or surgical instruments.
Antistatic properties: In operating room environments, they must prevent static electricity accumulation and reduce the risk of fire.
Strong and flexible:
Strong and tear-resistant, yet flexible and lightweight for easy laying and storage.
Sterilization:
The material itself must be able to withstand sterilization using gases (such as ethylene oxide) or pressurized steam.
AAMI plays a vital role in global healthcare. Its core functions include:
Standards Development:
AAMI is responsible for developing global consensus documents and performance standards to guide medical device manufacturers in producing products that meet safety and efficacy requirements. Although voluntary, these standards are widely adopted and serve as industry benchmarks.
Education and Training:
It provides education and training programs to ensure that healthcare professionals have the knowledge and skills necessary to properly operate, maintain, and apply modern medical technologies.
Advocacy and Collaboration:
It collaborates with government agencies, regulatory agencies (such as the US FDA), and other stakeholders to develop policies that encourage innovation and ensure patient safety.
Standards Development:
AAMI is responsible for developing global consensus documents and performance standards to guide medical device manufacturers in producing products that meet safety and efficacy requirements. Although voluntary, these standards are widely adopted and serve as industry benchmarks.
Education and Training:
It provides education and training programs to ensure that healthcare professionals have the knowledge and skills necessary to properly operate, maintain, and apply modern medical technologies.
Advocacy and Collaboration:
It collaborates with government agencies, regulatory agencies (such as the US FDA), and other stakeholders to develop policies that encourage innovation and ensure patient safety.
OUR FACTORY
Headquarter and Production Plants:
We are proud to serve healthcare providers, clinics, and distributors across Asia, Europe, the Middle East, and the Americas. Our products help protect medical staff and patients in critical environments—every single day.
460 +
Professional and Experienced staffready to help you
Client Testimonials
One-time Use. Long-term Trust
Professionals Worldwide

Exploring Anatomy Methods in the Lab
Most laboratory is a facility that provides controlled conditions in which…

A Laboratory Manual for Contemporary Specimen Gathering
Most laboratory is a facility that provides controlled conditions in which…

A Guide for Remote Patient Monitoring in a Lab.
Most laboratory is a facility that provides controlled conditions in which…

We will discuss the progress of our research.
Most laboratory is a facility that provides controlled conditions in which…




























